We have completed the NCERT/CBSE Solutions chapter-wise for Class 9 Science Chapter 4 Structure of the Atom with Answers by expert subject teacher for latest syllabus and examination. Prepare effectively for the exam taking the help of the Class 9 Science NCERT Solutions PDF free of cost from here. Students also can take a free NCERT Solutions of Structure of the Atom. Each question has right answer Solved by Expert Teacher. Download the Science NCERT Solutions with Answers for Class 9 Science Pdf and prepare to help students understand the concept very well.
NCERT Solutions for Class 9 Science Chapter wise
Questions
Q1. What are the canal rays?
Answer: Canal rays are positively charged radiations that can pass through perforated cathode plate. These rays consist of positively charged particles known as protons.
Q2. If an atom contains one electron and one proton, will it carry any charge or not?
Answer: Since a proton is a positively charged particle and an electron is a negatively charged particle, the net charge becomes neutral as both the particles neutralizes each other.
Questions
Q1. On the basis of Thompson’s model of an atom, explain how the atom is neutral as a whole.
Answer: According to Thomson’s model of an atom
(i) An atom consists of a positively charged sphere and the electrons are embedded in it,
(ii) The negative and positive charges are equal in magnitude. So the atom is electrically neutral.
Q2. On the basis of Rutherford’s model of an atom, which subatomic particle is present in the nucleus of an atom?
Answer: The nucleus of atom is positively charged according to Rutherford’s model of an atom. All the protons in an atom are therefore, present in the nucleus.
Q3. Draw a sketch of Bohr’s model of an atom with three shells.
Answer:
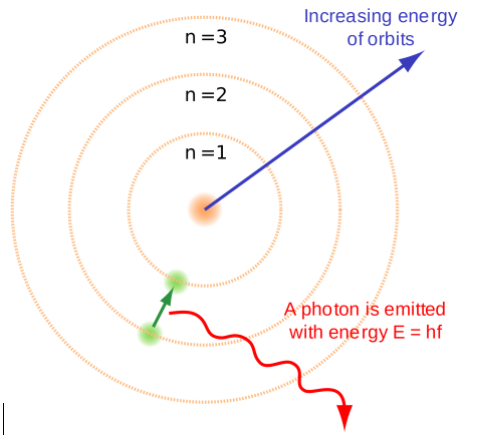
Q4. What do you think would be the observation if the ∝– particle scattering experiment is carried out using a foil of a metal other than gold?
Answer: If the α-scattering experiment is carried out using a foil of a metal rather than gold, there would be no change in the observation. In the α-scattering experiment, a gold foil was taken because gold is malleable and a thin foil of gold can be easily made. It is difficult to make such foils from other metals.
Questions
Q1. Name the three subatomic particles of an atom.
Answer: The three sub-atomic particles of an atom are :
- electron
- proton
- neutron.
Q2. Helium atom has an atomic mass of 4 u and two protons in its nucleus. How many neutrons does it have?
Answer: Helium atom has two neutrons. The mass of an atom is the sum of the masses of protons and neutrons present in its nucleus. Since helium atom has two protons, mass contributed by the two protons is (2 × 1) u = 2 u. Then, the remaining mass (4 − 2) u = 2 u is contributed by 2u /1u = 2 neutrons.
Questions
Q1. Write the distribution of electrons in Carbon and Sodium atoms.
Answer: The total number of electrons in a carbon atom is 6. The distribution of electrons in carbon atom is given by:
First orbit or K-shell = 2 electrons
Second orbit or L-shell = 4 electrons
Or, we can write the distribution of electrons in a carbon atom as 2, 4.
The total number of electrons in a sodium atom is 11. The distribution of electrons in sodium atom is given by:
First orbit or K-shell = 2 electrons
Second orbit or L-shell = 8 electrons
Third orbit or M-shell = 1 electron
Or, we can write distribution of electrons in a sodium atom as 2, 8, 1.
Q2. If K and L shells of an atom are full, then what would be the total number of electrons in the atom?
Answer: Maximum number of electron in K-shell = 22
Maximum number of electron in L-shell = 88
If K and L-shells of an atom are full,
then the total number of electrons in the atom would be (2 + 8) = 10
(2 + 8) = 10 electrons.
Questions
Q1. How will you find the valency of chlorine, sulphur and magnesium?
Answer: We know that an element’s valency refers to its proclivity for accepting or losing electrons in order to complete its octet and achieve a stable electronic state.
It is the smallest number of electrons that must be added or removed to entirely occupy an element’s outermost shell.
Mathematically, if an atom’s outermost shell contains 4 or fewer electrons, the element’s valency is equal to the number of electrons present in the outermost shell; if it contains more than 4, the valency is determined by subtracting the total number of electrons present in the outermost shell from 8.
Calculation of valency:
Valency of chlorine:
The electronic configuration of chlorine = 2, 8, 7.
Since, chlorine has 7 (more than 4) electrons in its outermost shell.
Therefore, valency of chlorine = 8 – number of electrons in outermost shell
= 8−7
= 1
Valency of Sulphur:
The electronic configuration of Sulphur = 2, 8,6.
Since, Sulphur has 6 (more than 4) electrons in its outermost shell.
Therefore, valency of chlorine = 8-number of electrons in outermost shell
= 8−6
= 2.
Valency of magnesium:
The electronic configuration of Magnesium = 2, 8, 2.
Since, magnesium has 2 (less than 4) electrons in its outermost shell.
Therefore, valency of magnesium= Number of electrons in its outermost shell
= 2
Questions
Q1. If the number of electrons in an atom is 8 and number of protons is also 8, then
(i) What is the atomic number of the atom? and
(ii) What is the charge on the atom?
Answer:

Q2. With the help of given Table, find out the mass number of oxygen and sulphur atom.
Table: Composition of Atoms of the First Eighteen Elements with Electron Distribution in Various Shells.
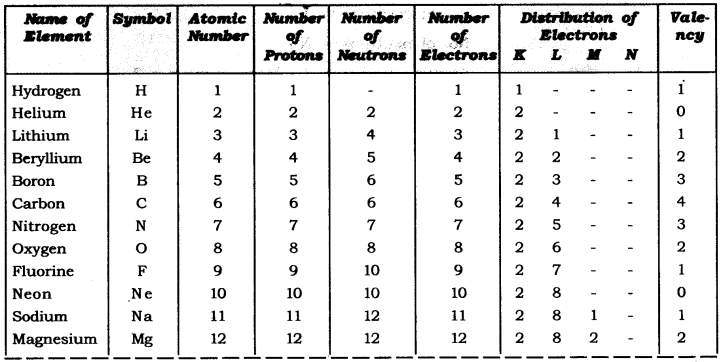
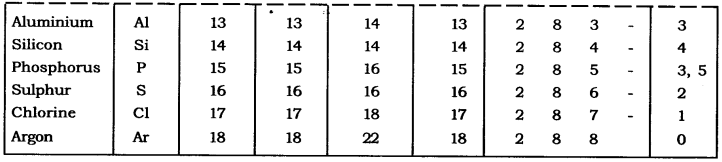
Answer:

Questions
Q1. For the symbol H, D and T, tabulate three subatomic particles found in each of them.
Answer:

Q2. Write the electronic configuration of any one pair of isotopes and isobar.
Answer:
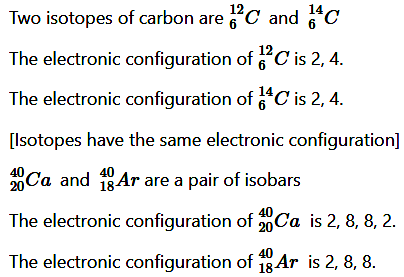
Exercises
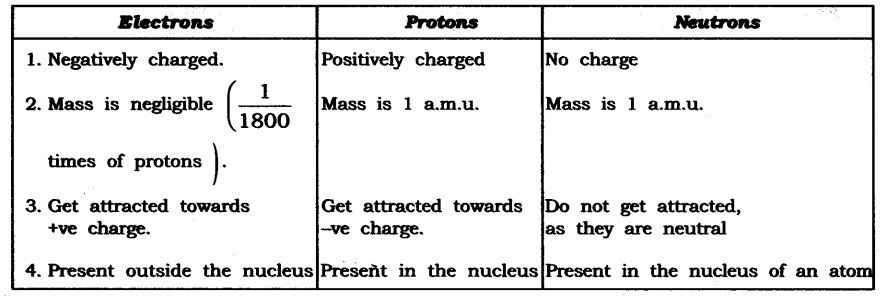
Q1. Compare the properties of electrons, protons and neutrons.
Answer:
Q2. What are the limitations of J.J.Thomson’s model of the atom?
Answer: The limitation of Thomson’s model are described below :
- Thomson’s model of an atom considers an atom to lae a sphere of uniform positive charge. Later researches particularly, Rutherford’s a-particle scattering experiment showed that an atom has a positively charged ‘core’ at its centre.
- According to Thomson’s atomic model, mass of an atom is considered to be uniformly distributed. Rutherford’s experiment showed that the entire mass of an atom is concentrated inside the core of the atom.
Q3. What are the limitations of Rutherford’s model of the atom?
Answer: According to Rutherford model, there is a positively charged centre in an atom called the nucleus and the electron present in nucleus revolve aroun it, in well- defined orbits. But, since an electron is charged particle. Thus, while revolving around the nucleus it lose energy and finally fall into the nucleus. If this were so, the atom should be highly unstable and hence matter would not exist in the form that we know.
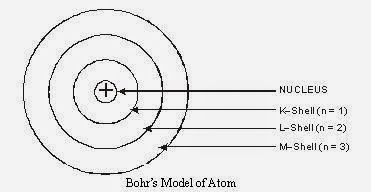
Q4. Describe Bohr’s model of the atom.
Answer:
- The atom consists of a small positively charged nucleus at its center.
- The whole mass of the atom is concentrated at the nucleus and the volume of the nucleus is much smaller than the volume of the atom.
- All the protons and neutrons of the atom are contained in the nucleus.
- Only certain orbits known as discrete orbits of electrons are allowed inside the atom.
- While revolving in these discrete orbits electrons do not radiate energy. These orbits or cells are represented by the letters K, L, M, N etc. or the numbers, n = 1, 2, 3, 4, . . as shown in below figure.
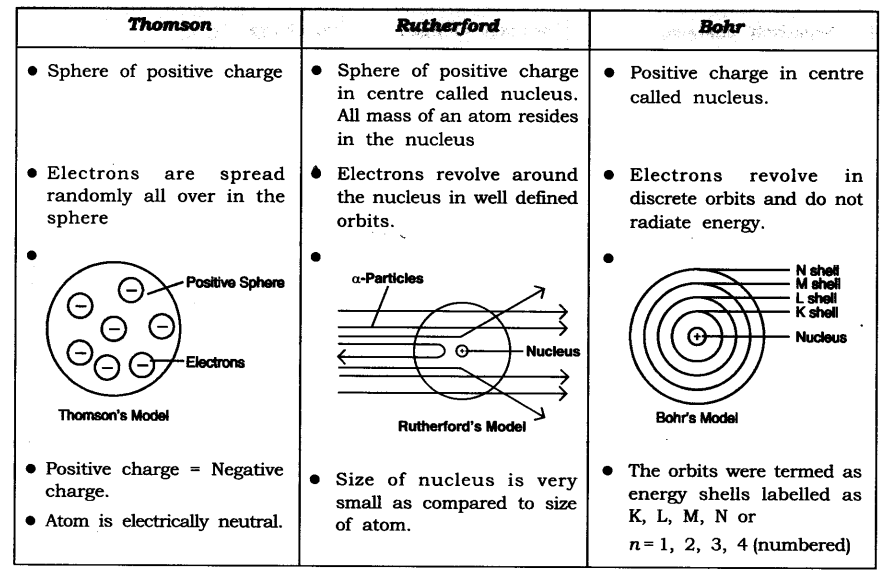
Q5. Compare all the proposed models of an atom given in this chapter.
Answer:
Q6. Summarize the rules for writing of distribution of electrons in various shells for the first eighteen elements.
Answer:
- Maximum number of electrons that can be accommodated in a shell is given by the formula: 2n², where n= 1, 2, 3…
- Maximum number of electrons in different shells are:
K shell – n=1 ; 2n² = 2(1)² = 2
L shell – n=2 ; 2n² = 2(2)² = 8
M shell – n=3 ; 2n² = 2(3)² = 18
N shell- n=4 ; 2n² = 2(4)² = 32
- The outermost orbit can be accommodated with 8 electrons at the maximum.
- The electrons are not taken in unless the inner shells are filled which are filled step-wise, hence the highest element has K-2; L-8 ; M-8 distribution of electrons.
Q7. Define valency by taking examples of silicon and oxygen.
Answer: Valency is the combining capacity of an atom.
Atomic number of oxygen = 8 Atomic number of silicon = 14 K L M
Electronic configuration of oxygen = 2 6 –
Electronic configuration of silicon =2 8 4
In the atoms of oxygen the valence electrons are 6 (i.e., electrons in the outermost shell). To fill the orbit, 2 electrons are required. In the atom of silicon, the valence electrons are 4. To fill this orbit 4 electrons are required.
Hence, the combining capacity of oxygen is 2 and of silicon is 4.
i.e., Valency of oxygen = 2
Valency of silicon = 4
Q8. Explain with examples (i) Atomic number, (ii) Mass number, (iii) Isotopes and (iv) Isobars. Give any two uses of isotopes.
Answer: (i) Atomic number:
Rutherford stated that the number of unit positive charges present in the nucleus of an atom is known as atomic number of the element It is denoted by the symbol Z.
Atomic no. (Z)= No. ofprotons = No. of electrons(ii) Mass number:
The mass number of an element may be defined as : the sum of the number of protons and neutrons present in the nucleus of its atom.
Mass number (A) = No. of protons + No. of neutrons
(iii) Isotopes:
Isotopes may be defined as the different atoms of the same element having same atomic number but different mass numbers.(iv) Isobars:
isobars may be defined as the atoms belonging to the different elements with same mass numbers but different atomic numbers.
Q9. Na+ has completely filled K and L shells. Explain.
Answer: An atom of Na has a total of 11 electrons. Its electronic configuration is 2, 8, 1. But, Na+ ion has one electron less than Na atom i.e., it has 10 electrons. Therefore, 2 electrons go to K-shell and 8 electrons go to L-shell, thereby completely filling K and L shells.
Q10. If bromine atom is available in the form of, say, two isotopes 35Br79 (49.7%) and 35Br81 (50.3%), calculate the average atomic mass of Bromine atom.
Answer: The atomic mass of an element is the mass of one atom of that element. Average atomic mass takes into account the isotopic abundance.
Isotope of bromine with atomic mass 79 u = 49.7%
Therefore, Contribution of 35Br79 to atomic mass = (79 × 49.7)/100
= 39.26 u
Isotope of bromine with atomic mass 81 u = 50.3%
Contribution of 35Br81 to the atomic mass of bromine = (81 × 50.3)/100
= 40.64u
Hence, average atomic mass of the bromine atom = 39.26 + 40.64 u = 79.9u
Q11. The average atomic mass of a sample of an element X is 16.2 u. What are the percentages of isotopes 8 X 16 and 8 X 18 in the sample?
Answer: Let the percentage of 8X16 be ‘a’ and that of 8X18 be ‘100-a’.
As per given data,
16.2u = 16 a / 100 + 18 (100-a) /100
1620 = 16a + 1800 – 18a
1620 = 1800 – 2a
a = 90%
Hence, the percentage of isotope in the sample 8X16 is 90% and that of
8X18 = 100-a = 100- 90=10%
Q12. If Z = 3, what would be the valency of the element? Also, name the element.
Answer: Given: Atomic number, Z = 3
The electronic configuration of the element = K-2; L-1, hence its valency = 1
The element with atomic number 3 is Lithium.
Q13. Composition of the nuclei of two atomic species X and Y are given as under
X Y
Protons = 6 6
Neutrons = 6 8
Give the mass numbers of X and Y. What is the relation between the two species?
Answer: Mass number of X: Protons + neutrons = 6+6 = 12
Mass number of Y: Protons + neutrons = 6+8 = 14
They are the same element as their atomic numbers are the same.
They are isotopes as they differ in the number of neutrons and hence their mass numbers.
Q14. For the following statements, write T for ‘True’ and F for ‘False’.
(a) J.J. Thomson proposed that the nucleus of an atom contains only nucleons.
(b) A neutron is formed by an electron and a proton combining together. Therefore, it is neutral.
(c) The mass of an electron is about 1 / 2000times that of proton.
(d) An isotope of iodine is used for making tincture iodine, which is used as a medicine.
Answer: (a) False
(b) False
(c) True
(d) False
Q15. Rutherford’s alpha-particle scattering experiment was responsible for the discovery of
(a) Atomic nucleus
(b) Electron
(c) Proton
(d) Neutron
Answer: (a) Atomic nucleus
Q16. Isotopes of an element have
(a) the same physical properties
(b) different chemical properties
(c) different number of neutrons
(d) different atomic numbers
Answer: (c) different number of neutrons
Q17. Number of valence electrons in Cl -ion are:
(a) 16
(b) 8
(c) 17
(d) 18
Answer: (b) 8
Page No: 56
Q18. Which one of the following is a correct electronic configuration of sodium?
(a) 2, 8
(b) 8, 2, 1
(c) 2, 1, 8
(d) 2, 8, 1
Answer: (d) 2, 8, 1
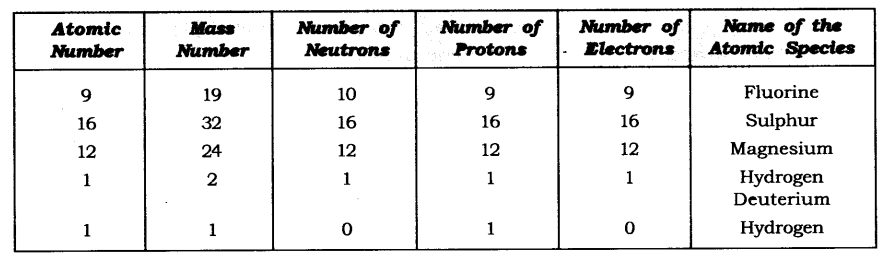
Q19. Complete the following table.
Answer:
We Think the given NCERT Solutions for class 9 Science book Chapter 4 Structure of the Atom with Answers Pdf free download will assist you. If you’ve got any queries regarding CBSE Class 9 Science Structure of the Atom NCERT Solutions with Answers, drop a comment below and that we will come back to you soons.