We have Provided the NCERT/CBSE Solutions chapter-wise for Class 10 Science Chapter 1 Chemical Reactions and Equations with Answers by expert subject teacher for latest syllabus and examination. Students can take a free NCERT Solutions of Chemical Reactions and Equations. Each question has right answer Solved by Expert Teacher.
CBSE Solutions Class 10 Science Chemical Reactions and Equations
Questions
Q1. Why should a magnesium ribbon be cleaned before burning in air?
Answer: A layer of magnesium oxide is already present on the surface of magnesium ribbon which does not allow burning of magnesium ribbon in air. So magnesium ribbon should be cleaned before burning in air.
Q2. Write the balanced equation for the following chemical reactions.
(i) Hydrogen + Chlorine → Hydrogen chloride
(ii) Barium chloride + Aluminium sulphate → Barium sulphate + Aluminium chloride
(iii) Sodium + Water → Sodium hydroxide + Hydrogen
Answer:
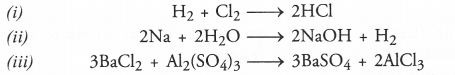
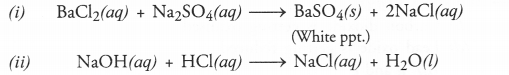
Q3. Write a balanced chemical equation with state symbols for the following reactions.
(i) Solutions of barium chloride and sodium sulphate in water react to give insoluble barium sulphate and the solution of sodium chloride.
(ii) Sodium hydroxide solution (in water) reacts with hydrochloric acid solution (in water) to produce sodium chloride solution and water.
Answer:
Page 10
Questions
Q1. A solution of a substance ‘X is used for white-washing:
(i) Name the substance ‘X’ and write its formula.
Answer: (i) The substance ‘X’ is quick lime or calcium oxide.
(ii) Write the reaction of the substance ‘X’ named in (i) above with water.
Answer:
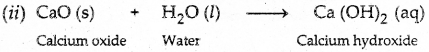
Q2. Why is the amount of gas collected in one of the test tubes in activity 1.7 double of the amount collected in the other ? Name this gas.
Answer: In activity 1.7, gas collected in one of the test tubes is double of the amount collected in the other because water gets hydrolysed to release H2 and O2 gas. Here, after electrolysis two molecules of Hydrogen and one molecule of oxygen gas is released, hence the amount of Hydrogen collected would be double than that of oxygen.
Page No 13
Questions
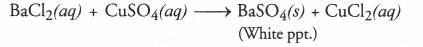
Q1. Why does the colour of copper sulphate solution change when an iron nail is dipped in it?
Answer: Iron nail acquires a brown coating of copper as a result of the displacement reaction in which iron has displaced copper from copper sulphate solution. The solution becomes light green.
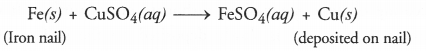
Q2. Give an example of a double displacement reaction other than the one given in Activity 1.10.
Answer:
Q3. Identify the substances that are oxidised and the substances that are reduced in the following reactions.
Answer:
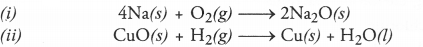
Answer: (i) In this reaction, sodium (Na) is oxidised to sodium oxide (Na2O). This means that oxygen (O2) has been reduced.
(ii) In this reaction, hydrogen (H2) is oxidised to water (H2O) while copper (II) oxide (CuO) is reduced to copper (Cu).
Page 14
EXERCISES
Q1. Which of the statements about the reaction below are incorrect?
2PbO(s) + C(s) → 2Pb(s) + CO2(g)
(a) Lead is getting reduced.
(b) Carbon dioxide is getting oxidised.
(c) Carbon is getting oxidised.
(d) Lead oxide is getting reduced.
(i) (a) and (b)
(ii) (a) and (c)
(iii) (d), (b) and (c)
(iv) all
Answer: (i) (a) and (b)
Q2. Fe2O3 + 2Al → Al2O3 + 2Fe
The above reaction is an example of a
(a) combination reaction.
(b) double displacement reaction.
(c) decomposition reaction.
(d) displacement reaction.
Answer: (d) displacement reaction.
Q3. What happens when dilute hydrochloric add is added to iron fillings? Tick the correct answer.
(a) Hydrogen gas and iron chloride are produced.
(b) Chlorine gas and iron hydroxide are produced.
(c) No reaction takes place.
(d) Iron salt and water are produced.
Answer: (a) Hydrogen gas and iron chloride are produced.
Q4. What is a balanced chemical equation? Why should chemical equations be balanced?
Answer: A balanced equation is the one in which number of different atoms on both the reactant and product sides are equal. Balancing chemical equation is necessary for the reaction should obey The Law of Conservation of energy. Balancing the chemical equation has no defined method and is purely a trial and error attempt.
Q5. Translate the following statements into chemical equations and then balance them.
(a) Hydrogen gas combines with nitrogen to form ammonia.
Answer:
H2 (g) + N2(g) → NH3 (g)
Step I. Look more closely the number of atoms of different elements present in the unbalanced chemical equation.
| Element | No. of atoms in Reactants | No. of atoms in Products |
| H | 2 | 3 |
| N | 2 | 1 |
Step II. It is often convenient to start with the compound that contains maximum number of atoms, whether a reactant or a product. So we select NH3 and the element hydrogen in it. There are three hydrogen atoms on the right and only two hydrogen atoms on the left.
| Atoms of Hydrogen | In reactants | In Products |
| Initial | 2 (in H2) | 3 (in NH3) |
| To balance | 2 × 3 | 3 × 2 |
Now the partly balanced equation becomes as follows :
3H2 + N2 → 2NH3
Step III. But nitrogen in the above equation is automatically balanced. So the balanced equation will be
3 H2 + N2 → NH3
The above equation balanced because it contains the equal no. of atoms of hydrogen and nitrogen on both sides.
(b) Hydrogen sulphide gas bums in air to give water and sulpur dioxide.
Answer:
H2S + O2 → H2O + H2O
The above equation can be balanced by algebraic sum method.
Let ‘a’, ‘b’, ‘c’ and ‘d’ are the no. of molecules of ‘H2S’, ‘O2‘, ‘H2O’ and ‘SO2‘ respectively in the balanced chemical equation. So the above equation can be written as
a H2S + b O2 → c H2O + d SO2
Now in a balanced chemical equation.
L.H.S. R.H.S.
Hydrogen 2 a= 2c …(i)
Sulphur a = d …(ii)
Oxygen 2b = c + 2d ….(iii)
Let a = 1 …..(iv)
So from equations (i) and (iv)
2 × 1 = 2 c
⇒ c = 1
From equation (iii)
2b = c + 2d
2b = 1 + 2 × 1
⇒ 2b = 3
⇒ b = 32
Now by putting the value of a, b, c and d in the given equation, we get
1 × H2S + 32O2 → 1 × H2O + 1 × SO2
OR 2 H2S + 3O2 → 2 H2O + 2SO2
(c) Barium chloride reacts with aluminium sulphate to give aluminium chloride and a precipitate of barium sulphate.
Answer:
The symbol equation for the reaction is :
BaCl2 + Al2(SO4)3 ———-> A1Cl3 + BaSO4
The balancing of equation is done in the following steps :
Step I: Let us count the number of atoms of all the elements on both sides of the equation.
| Elements | No. of atoms of reactants (L.H.S.) | No. of atoms of products (R.H.S.) |
| Ba | 1 | 1 |
| A1 | 2 | 1 |
| Cl | 2 | 3 |
| S | 3 | 1 |
| O | 12 | 4 |
A simple look at the equation reveals that only Ba atoms are equal on both sides. The rest of the atoms are to be balanced. It is done as follows :
Step II: In order to equate the number of Al atoms, put coefficient 2 before AlCl3 on the product side.
BaCl2 + Al2(SO4)3 ———-> 2AICl3 + BaSO4
Step III: In order to balance Cl atoms, put coefficient 3 before BaCl2 on the reactant side.
3BaCl2 + Al2(SO4)3 ———–> 2AlCl3 + BaSO4
Step IV: To balance Ba atoms, put coefficient 3 before BaSO4 on the product side.
3BaCl2 + Al2(SIO4)3 ———-> 2AlCl3 + 3BaSO4
Step V: On inspection, the number of S and O atoms on both sides of the equation are also found to be equal. Thus, the equation is in balanced form.
(d) Potassium metal reacts with water to give potassium hydroxide and hydrogen gas.
Answer:
K + H2O → KOH + H2
Step I.
| Element | No. of atoms in Reactants | No. of atoms in Products |
| K | 1 | 1 |
| H | 2 | 3 |
| O | 1 | 1 |
Step II: Now select ‘KOH’ in the reactant side because it contains the maximum no. of elements.
K + 2H2O → 2 KOH + H2
Step III: Balance potassium metal in the above partially balanced equation by multiplying ‘2’ in the potassium i.e. reactant side.
2K + 2H2O → 2KOH + H2
Q6. Balance the following chemical equations.
(a) HNO3 + Ca(OH)2 → Ca(NO3)2 + H2O
(b) NaOH + H2SO4 → Na2SO4 + H2O
(c) NaCl + AgNO3 → AgCl + NaNO3
(d) BaCl2 + H2SO4 → BaSO4 + HCl
Answer:
(i) 2HNO3 + Ca(OH)2 → Ca(NO3)2 + 2H2O
(ii) 2NaOH + H2SO4 → Na2SO4 + 2H2O
(iii) NaCl + AgNO3 → AgCl + NaNO3
(iv) BaCl2 + H2SO4 → BaSO4 + 2HCl
Q7. Write the balanced chemical equations for the following reactions.
(a) Calcium hydroxide + Carbon dioxide → Calcium carbonate + Water
Answer: Ca(OH)2 + CO2 → CaCO3 + H2O
(b) Zinc + Silver nitrate → Zinc nitrate + Silver
Answer: Zn + 2AgNO3 → Zn(NO3)2 + 2Ag
(c) Aluminium + Copper chloride → Aluminium chloride + Copper
Answer: 2Al + 3CuCl2 → 2AlCl3 + 3Cu
(d) Barium chloride + Potassium sulphate → Barium sulphate + Potassium chloride
Answer: BaCl2 + K2SO4 → BaSO4 + 2KCl
Q8. Write the balanced chemical equation for the following and identify the type of reaction in each case.
(a) Potassium bromide (aq) + Barium iodide (aq) → Potassium iodide (aq) + Barium bromide(s)
Answer: 2KBr (aq) + BaI2 (aq) → 2KI (aq) + BaBr2 (s): Double displacement reaction
(b) Zinc carbonate (s) → Zinc oxide (s) + Carbon dioxide (g)
Answer: ZnCO3 (s) → ZnO (s) + CO2 (g): Decomposition reaction
(c) Hydrogen (g) + Chlorine (g) → Hydrogen chloride (g)
Answer: H2 (g) + Cl2 (g) → 2HCl (g): Combination reaction
(d) Magnesium (s) + Hydrochloric acid (aq) → Magnesium chloride (aq) + Hydrogen (g)
Answer: Mg (s) + 2HCl (aq) → MgCl2 (aq) + H2 (g) : Displacement Reaction
Q9. What does one mean by exothermic and endothermic reactions? Give examples.
Answer: Chemical reactions that release energy in the form of heat, light, or sound are called exothermic reactions.
Example: C (g) + O2 (g) → CO2 + Heat Energy
Reactions that absorb energy or require energy in order to proceed are called endothermic reactions.
Example:
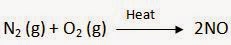
Q10. Why is respiration considered an exothermic reaction? Explain.
Answer: Respiration is considered as an exothermic reaction because in respiration oxidation of glucose takes place which produces large amount of heat energy.
C6H12O6 (aq) + 6O2 (g) → 6CO2 (g) + 6H2O (l) + Energy
Q11. Why are decomposition reactions called the opposite of combination reactions? Write equations for these reactions.
Answer:
Combination reaction may be defined as the reaction in which two or more substances combine under suitable conditions to form a new substance.
For example:

A decomposition reaction may be defined as the reaction in which a single substance decomposes or splits into two or more substances under suitable conditions.
For example:
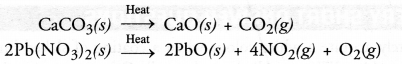
It may be concluded that a certain substance is formed or synthesised in combination reaction and it breaks or splits in decomposition reaction. Therefore, the two reactions oppose each other.
Q12. Write one equation each for decomposition reactions where energy is supplied in the form of heat, light or electricity.
Answer:
Write equation each for decomposition reactions where energy is supplied in the form of heat, light or electricity.
Answer:
In a decomposition reaction a single reactant breaks down to give two or more simpler products. These reactions need energy in different forms to proceed.
The decomposition of CaCO3 (s) takes place by supplying energy in the form of heat.
CaCO3(s)→ΔCaO(s)+CO2(g)
In the presence of sunlight white silver chloride decomposes and turns grey because silver metal is formed.![]()
Water decomposes into H, and O, by passing electricity in an electrolytic cell.
2H2O(l)→ Electricity 2H2(g)O2(g)
Q13. What is the difference between displacement and double displacement reactions? Write equations for these reactions.
Answer: n a displacement reaction, one element takes the place of another in a compound. For example,
Fe(s) + CuSO4(aq) ➝ FeSO4(aq) + Cu(s)
In a double displacement reaction, one com-ponent each of both the reactants get exchanged to form the products. For example,
HCl(aq) + NaOH(aq) ➝ NaCl(aq) + H2O(l)
Q14. In the refining of silver, the recovery of silver from silver nitrate solution involved displacement by copper metal. Write down the reaction involved.
Answer: Cu(s) + 2AgNO3(aq) → Cu(NO3)2(aq) + 2Ag(s)
Q15. What do you mean by a precipitation reaction? Explain by giving examples.
Answer: When two solutions containing soluble salts are combined, a double displacement reaction takes place in which the ions are exchanged between the compounds. When one of such compounds formed is in solid form (that is insoluble in aqua) then it settles down at the bottom of the container. This solid is known as the precipitate and the respective reaction is termed as the precipitation reaction. Few examples of precipitation reactions are:
CdSO4(aq) + K2S(aq) → CdS(s) + K2SO4(aq)
2NaOH(aq) + MgCl2(aq) → 2NaCl(aq) + Mg(OH)2(s)
Q16. Explain the following in terms of gain or loss of oxygen with two examples eac.?
(a) Oxidation
(b) Reduction
Answer: Oxidation Reaction: It is a chemical reaction in which gain of oxygen or loss of hydrogen takes place.

Reduction Reaction: It is a chemical reaction in which loss of oxygen or gain of hydrogen takes place.

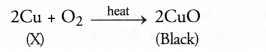
Q17. A shining brown coloured element ‘X’ on heating in’ air becomes black in colour. Name the element ‘X’ and the black coloured compound formed.
Answer: The element ‘X’ is copper and the black coloured compound is copper (II) oxide also known as cupric oxide.
Q18. Why do we apply paint on iron articles?
Answer: Iron articles are painted to prevent them from rusting. When left unpainted, the metal surface comes in contact with the atmospheric oxygen and in the presence of moisture it from Iron(III) oxide. But if painted the surface does not come in contact with moisture and air thus preventing Rusting.
Q19. Oil and Fat containing food items are flushed with Nitrogen. Why?
Answer: The main purpose of flushing Nitrogen into food packets that contain oil and fat items is to prevent Rancidity which occurs when the oil or fat reacts with the oxygen letting out an unpleasant smell and taste. Therefore, by flushing Nitrogen, an unreactive surrounding is created thus preventing rancidity.
Q20. Explain the following terms with one example each.
(a) Corrosion
Answer: Corrosion : The process of slowly eating away of the metals when they are exposed by air, moisture or other atmospheric gases, resulting into the formation of compounds such as oxides, sulphides, carbonates etc. is called corrosion.
Iron is corroded when exposed by moisture or air. Corrosion of iron is also known as rusting. Rust is mainy hydrated iron (III) oxide i.e. Fe2O3. xH2O.
Rusting weakness the structure of car bodies, bridges, iron railings and other iron articles. Rusting of iron is a serious problem. Every year a large amount of money is spent to replace damaged iron.
(b) Rancidity
Answer: Rancidity: When fats and oils are oxidised they become rancid and their smell and taste change. Generally special type of substances i.e., antioxidants are added to fatty foods to prevent oxidation. At home oxidation of food can be slowed down by keeping it in the refrigerator. Keeping and food in air-tight containers can also help.
We Think the given NCERT Solutions for class 10 Science Chapter 1 Chemical Reactions and Equations with Answers Pdf free download will assist you. If you’ve got any queries regarding CBSE Class 10 Science Chemical Reactions and Equations NCERT Solutions with Answers, drop a comment below and that we will come back to you soons.
