We have completed the NCERT/CBSE Solutions chapter-wise for Class 9 Science Chapter 1 Matter in Our Surroundings with Answers by expert subject teacher for latest syllabus and examination. Prepare effectively for the exam taking the help of the Class 9 Science NCERT Solutions PDF free of cost from here. Students also can take a free NCERT Solutions of Matter in Our Surroundings. Each question has right answer Solved by Expert Teacher. Download the Science NCERT Solutions with Answers for Class 9 Science Pdf and prepare to help students understand the concept very well.
NCERT Solutions for Class 9 Science Chapter wise
Page No. 3
In Text Questions
Q1. Which of the following are matter?
Chair, air, love, smell, hate, almonds, thought, cold, cold drink, smell of perfume.
Answer: Chair, air, almonds and cold drink
Q2. Give reasons for the following observation:
The smell of hot sizzling food reaches you several metres away, but to get the smell from cold food you have to go close.
Answer: Particles in the air, if fueled with higher temperatures, acquire high kinetic energy which aids them o move fast over a stretch. Hence the smell of hot sizzling food reaches a person even at a distance
of several meters.
Q3. A diver is able to cut through water in a swimming pool. Which property of matter does this observation show?
Answer: A diver is able to cut through water in a swimming pool. This shows that the particles of water have intermolecular space and has less force of attraction.
Q4. What are the characteristics of particles of matter?
Answer: Matter is anything that occupies space and has some mass. There are three states of matter called Solid, Liquid and Gas.
The main characteristics of matter can be described as follows:
- Particles of matter have space between them and the order of spacing is highest in gas after that liquid and solid have very less space between their particles.
- Particles of matter are continuously moving in all the three states of matter.
- Particles of matter attract each other with strong forces which help them to bind with each other. In solid particles attraction is very high whereas in liquid it is low and in gases it is quite low.
Questions
Q1. The mass per unit volume of a substance is called density (density = mass/volume).
Arrange the following in order of increasing density – air, exhaust from chimney, honey, water, chalk, cotton, and iron.
Answer: The given substances in the increasing order of their densities can be represented as:
Air < Exhaust from chimney < Cotton < Water < Honey < Chalk < Iron
Q2. (a) Tabulate the differences in the characteristics of states of matter.
Answer:
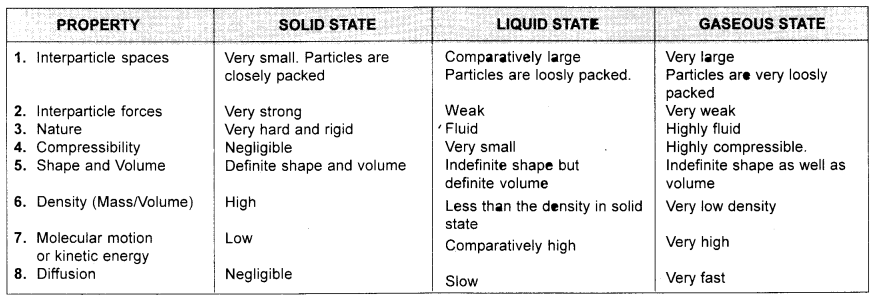
(b) Comment upon the following: rigidity, compressibility, fluidity, filling a gas container, shape, kinetic energy, and density.
Answer: (b) (i) Rigidity : It is the property which helps a substance to retain its shape when force is applied to it. Splids are rigid while gases and liquids are not.
(ii) Compressibility : The property due to which the particles of matter can be compressed or reduced in volume by applying force or pressure. Gases are highly compressible.
(iii) Fluidity: It is the tendency of a substance to flow. Liquids and gases possess fluidity while solids are rigid.
(iv) Filling a gas container : The molecules of a gas move in all directions and due to negligible inter particle force of attraction can fill the container.
(v) Shape: Solids have definite shape whereas liquids take the shape of the container in which they are placed and gases do not have any shape.
(vi) Kinetic energy: It is the energy possessed by the particles due to their motion. The particles of a gas have maximum kinetic energy due to free motion of gas particles in all directions. Solids have minimum kinetic energy due to least movement of particles.
(vii) Density : Density is the mass of a substance per unit volume. Solids have highest density since their molecules are closely packed.
Q3. Give reasons
a) A gas fills completely the vessel in which it is kept.
Answer: This is because the gases have large inter-particle spaces and the inter-particle attraction force is very weak. The gaseous particle can move freely into the empty spaces of another gas as particles of gas have very high kinetic energy. This property of gases is called diffusion. A gas can mix with any other gas easily and move rapidly from one part of vessel to another part of vessel. Therefore, gas particles can spread easily and fill the whole vessel.
b) A gas exerts pressure on the walls of the container.
Answer: Particles of gas move randomly in all directions at high speed. As a result, the particles hit each other and also hit the walls of the container with a force. Therefore, gas exerts pressure on the walls of the container.
c) A wooden table should be called a solid.
Answer: There is a distinct contour and volume to the hardwood table. The wood particles are tightly packed. They do not conform to the container’s shape. As a result, the solid features of a hardwood table are satisfied.
d) We can easily move our hand in the air but to do the same through a solid block of wood we need a karate expert.
Answer: The boundaries between air particles are quite loose. They are a long way apart and have a lot of space between them. As a result, we may move our hands freely in the air. The particles in a solid block, on the other hand, are bound together by a strong force of attraction. As a result, there is either some or no space between them. As a result, we’ll require a karate expert.
Q4. Liquids generally have a lower density than solids. But you must have observed that ice floats on water. Find out why.
Answer: Ice is a solid but its density is lower than water due to its structure. The molecules in ice make a cage like structure with lot of vacant spaces, this makes ice float on water.
Questions
Q1. Convert the following temperature to Celsius scale:
(a) 300K
(b) 573K
Answer:
(a) (300 – 273) = 27°C
(b) (573 – 273) = 300°C.
Q2. What is the physical state of water at:
(a) 250°C
(b) 100°C ?
Answer: (a) 250ºC = gas
(b) 100°C liquid as well as gas
Q3. For any substance, why does the temperature remain constant during the change of state?
Answer: The temperature remains constant during the change of state because the heat supplied during the change is used up in overcoming the inter molecular forces between the particles of the state.
Q4. Suggest a method to liquefy atmospheric gases.
Answer: In liquid, the particles are closer to each other and have lower kinetic energy than gases. When pressure is applied to gas particles they move closer and loose K.E. and thus change to their liquid state. It is possible to liquefy any gas. i.e., convert gas into liquid by increasing the pressure and decreasing the temperature.
Questions
Q1. Why does a desert cooler cool better on a hot dry day?
Answer: A desert cooler increases the humidity of the surrounding air. The water particles in the air take the heat from the surrounding objects and evaporate. In hot and dry days the moisture level is very low in the atmosphere which increases the rate of evaporation. Because of faster evaporation, the cooler works well. That’s why desert cooler cools better on a hot dry day.
Q2. How does the water kept in an earthen pot (matka) become cool during summer?
Answer: An earthen pot is porous in nature. These tiny pores facilitate penetration of water and hence their evaporation from the pot surface. The process of evaporation requires energy which is contributed by water in the pot as a result of which water turns cooler.
Q3.Why does our palm feel cold when we put on some acetone or petrol or perfume on it?
Answer: Acetone, petrol or perfume evaporate when they come into contact with air. The evaporation causes cooling sensation in our hands.
Q4. Why are we able to sip hot tea or milk faster from a saucer rather than a cup?
Answer: This can also be explained on the basis of rate of evaporation as we know that evaporation produces a cooling effect and evaporation depends on the surface area, larger the surface area higher the evaporation. As in saucer the area is larger as compared to cup so evaporation will be high in case of greater surface area. Thus, we can say that liquid cools faster in a saucer than in a cup and due to this reason, we are able to sip hot tea or milk faster from a saucer than a cup.
Q5. What type of clothes should we wear in summer?
Answer: We should wear cotton clothes in summers. During summers, we sweat more. On the other hand, cotton is a good absorber of water. Thus, it absorbs sweat from our body and exposes the liquid to the atmosphere, making evaporation faster. During this evaporation, particles on the surface of the liquid gain energy from our body surface, making the body cool.
Page No. 12
Exercises
Q1. Convert the following temperature to Celsius scale.
(a) 293K (b) 470K
Answer: (a) We know that, temperature in °C = K – 273 = 293 – 273 = 20°C
(b) °C = K – 273 = 470 – 273 = 197°C
Q2.Convert the following temperatures to the Kelvin scale.
(a) 25°C (b) 373°C
Answer: (a) We know that temperature in K = °C + 273 K = 25° + 273 = 298 K
(b) K = °C + 273 = 373 + 273 = 646 K
Q3. Give reason for the following observations:
(a) Naphthalene balls disappear with time without leaving any solid.
Answer: Naphthalene shows the property of sublimation. Evaporation of naphthalene takes place easily and so it disappears during course of time without leaving any solid.
(b) We can get the smell of perfume while sitting several metres away.
Answer: Perfumes have high degree of vaporisation and its vapour diffuses into the air easily. Therefore, we can get the smell of perfume sitting several metres away.
Q4. Arrange the following in increasing order of forces of attraction between the particles – water, sugar, oxygen.
Answer: Oxygen (gas) < water (liquid) < sugar (solid)
Q5. What is the physical state of water at –
(a) 25°C (b) 0°C (c) 100°C?
Answer: (a) At 25°C, the water will be in liquid form (normal room temperature)
(b) At 0°C, the water is at its freezing point, hence both solid and liquid phases are observed.
(c) At 100°C, the water is at its boiling point, hence both liquid and gaseous state of water (water
Q6.Give two reasons to justify –
(a) Water at room temperature is a liquid.
Answer: (a) Water at room temperature is a liquid because its freezing point is 0°C and boiling point is 100°C.
(b) An iron almirah is a solid at room temperature.
Answer: (b) An iron almirah is a solid at room temperature because melting point of iron is higher than room temperature.
Q7. Why is ice at 273K more effective in cooling than water at the same temperature?
Answer: Here condition given that both ice and water are at same temperature i.e. 273 K. But ice at 273 K has less energy as compared to water this can be explained on the basis of latent heat of fusion which is possessed by water as an additional energy but ice does not have such type of energy. Therefore, we can say that at 273 K ice is more effective in cooling as compared to water.
Q8. What produces more severe burns, boiling water or steam?
Answer: Steam has more energy than boiling water. It possesses the additional latent heat of vaporisation. Therefore, burns produced by steam are more severe than those produced by boiling water.
Q9. Name A, B, C, D, E and F in the following diagram showing a change in its state.
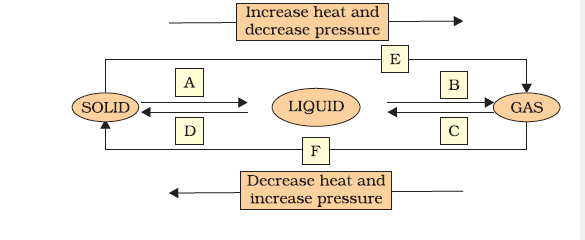
Answer:
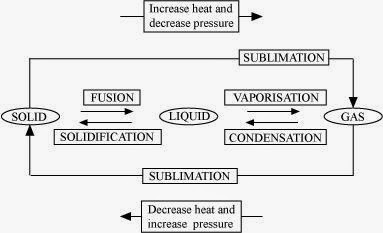
We Think the given NCERT Solutions for class 9 Science book Chapter 1 Matter in Our Surroundings with Answers Pdf free download will assist you. If you’ve got any queries regarding CBSE Class 9 Science Matter in Our Surroundings NCERT Solutions with Answers, drop a comment below and that we will come back to you soons.
