We have completed the NCERT/CBSE Solutions chapter-wise for Class 9 Science Chapte2 Is Matter Around Us Pure with Answers by expert subject teacher for latest syllabus and examination. Prepare effectively for the exam taking the help of the Class 9 Science NCERT Solutions PDF free of cost from here. Students can take a free NCERT Solutions of Is Matter Around Us Pure. Each question has right answer Solved by Expert Teacher. Download the Science NCERT Solutions with Answers for Class 9 Science Pdf and prepare to help students understand the concept very well.
NCERT Solutions for Class 9 Science Chapter wise
Page No: 15
Questions
Q1. What is meant by a pure substance?
Answer: A material that is composed of only one type of particles is called pure substance. All the constituent particles of a pure substance have same chemical nature.
Q2. List the points of differences between homogeneous and heterogeneous mixtures.
Answer:
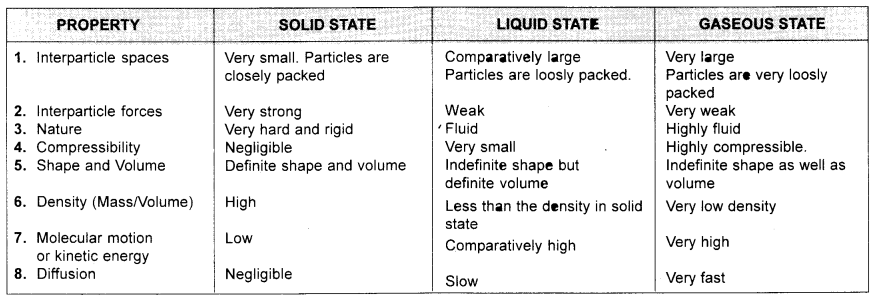
Questions
Q1. Differentiate between homogeneous and heterogeneous mixtures with examples.
Answer: A homogeneous mixture is a mixture having a uniform composition throughout the mixture. For example, mixtures of salt in water, sugar in water, copper sulphate in water, iodine in alcohol, alloy, and air have uniform compositions throughout the mixtures.
On the other hand, a heterogeneous mixture is a mixture having a non-uniform composition throughout the mixture. For example, composition of mixtures of sodium chloride and iron fillings, salt and sulphur, oil and water, chalk powder in water, wheat flour in water, milk and water are not uniform throughout the mixtures.
Q2. How are sol, solution and suspension different from each other?
Answer:

Q3. To make a saturated solution, 36 g of sodium chloride is dissolved in 100 g of water at 293 K. Find its concentration at this temperature.
Answer:

Questions
Q1. How will you separate a mixture containing kerosene and petrol (difference in their boiling points is more than 25°C), which are miscible with each other?
Answer: The separation can be done by applying process of simple distillation. Both are miscible liquids. Since the difference in the boiling points is less than 25 K, the separation can be done with the help of fractional distillation technique. Petrol with less boiling point distils first leaving behind kerosene in the distillation from flask.
Q2. Name the techniques used to separate the following:
(a) Butter from curd.
(b) Salt from seawater
(c) Camphor from salt
Answer: (i) Butter from curd can be separated by the technique of centrifugation.
(ii) Salt from sea water can be separated by the technique of crystallisation or evaporation.
(iii) Camphor is sublimable but salt is not. So, camphor can be separated from salt by sublimation.
Q3. What type of mixtures are separated by the technique of crystallization?
Answer: By the technique of crystallization, pure solids are separated from impurities. For example, salt obtained from sea is separated from impurities; crystals of alum (Phitkari) are separated from impure samples.
Questions
Q1. Classify the following as chemical or physical changes:
- Cutting of trees
- Melting of butter in a pan
- Rusting of almirah
- Boiling of water to form steam
- Passing of electric current through water and water breaking into hydrogen and oxygen gases.
- Dissolving common salt in water
- Making a fruit salad with raw fruits, and
- Burning of paper and wood
Answer:

Q2. Try segregating the things around you as pure substances and mixtures.
Answer: Pure substances—Water, bread, sugar and gold.
Mixtures—Steel, plastic, paper, talc, milk and air.
Exercise
Q1. Which separation techniques will you apply for the separation of the following?
(a) Sodium chloride from its solution in water.
(b) Ammonium chloride from a mixture containing sodium chloride and ammonium chloride.
(c) Small pieces of metal in the engine oil of a car.
(d) Different pigments from an extract of flower petals.
(e) Butter from curd.
(f) Oil from water.
(g) Tea leaves from tea.
(h) Iron pins from sand.
(i) Wheat grains from husk.
(j) Fine mud particles suspended in water.
Answer: (a) Evaporation
(b) Sublimation
(c) Filtration
(d) Chromatography
(e) Centrifugation
(f) Separating funnel
(g) Filtration
(h) Magnetic separation
(i) Winnowing/ sedimentation
(j) Decantation and filtration
Q2. Write the steps you would use for making tea. Use the words solution, solvent, solute, dissolve, soluble, insoluble, filtrate, and residue.
Answer: (a) Into a vessel, add a cup of milk which is the solvent, supply it with heat.
(b) Add tea powder or tea leaves to the boiling milk, which acts as a solute. Continue to heat
(c) The solute i.e., the tea powder remains insoluble in the milk which can be observed while it is still boiling.
(d) At this stage, add some sugar to the boiling solution while stirring
(e) Sugar is a solute but is soluble in the solvent
(f) Continuous stirring causes the sugar to completely dissolve in the tea solution hence reaching saturation.
(g) Once the raw smell of tea leaves is vanished and tea solution is boiled enough, take the solution off the heat, filter or strain it to separate tea powder and the tea solution. The insoluble tea powder remains as a residue while the solute (sugar) and the solvent (essenced milk solution) strain through the filter medium which is collected as the filtrate.
Q3. Pragya tested the solubility of three different substances at different temperatures and collected the data as given below( results are given in the following table, as grams of substance dissolved in 100 grams of water to form a saturated solution).
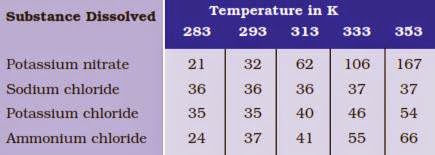
(a) What mass of potassium nitrate would be needed to produce a saturated solution of potassium nitrate in 50 grams of water at 313 K?
Answer: (a) At-313 K, in the saturated solution
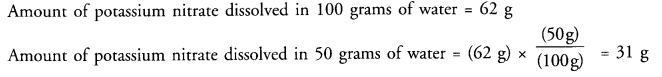
(b) Pragya makes a saturated solution of potassium chloride in water at 353 K and leaves the solution to cool at room temperature. What would she observe as the solution cools? Explain.
Answer: When the saturated solution prepared at 353 K is cooled to room temperature (about 298 K) the solubility of potassium chloride in water will decrease. It will slowly start separating as a crystalline white residue at the bottom of the container.
(c) Find the solubility of each salt at 293 K. What salt has the highest solubility at this temperature?
Answer: The solubility of the salt in a water is defined as :
the maximum amount of the salt which is dissolved in 100 g of water (or any other solvent) to form a saturated solution at a given temperature
In the light of this, at 293 K

(d) What is the effect of change of temperature on the solubility of a salt?
Answer: The solubility of a salt increases with temperature.
Q4. Explain the following giving examples:
(a) Saturated solution
(b) Pure substance
(c) Colloid
(d) Suspension
Answer: (a) Solution in which no more solute can be dissolved at a particular temperature is known as saturated solution. For example in aqueous solution of sugar no more sugar can be dissolved at room temperature.
(b) A pure substance is a substance consisting of a single type of particles i.e., all constituent particles of the substance have the same chemical properties. For example water, sugar, salt etc.
(c) A colloid is a heterogeneous mixture whose particles are not as small as solution but they are so small that cannot be seen by naked eye. When a beam of light is passed through a colloid then the path of the light becomes visible. For example milk, smoke etc.
(d) A suspension is a heterogeneous mixture in which solids are dispersed in liquids. The solute particles in suspension do not dissolve but remain suspended throughout the medium. For example Paints, Muddy water chalk water mixtures etc.
Q5. Classify each of the following as a homogeneous or heterogeneous mixture.
Soda water, wood, air, soil, vinegar, filtered tea
Answer: Homogeneous: Soda water, vinegar, filtered tea.
Heterogeneous: Wood, air, soil.
Q6. How would you confirm that a colourless liquid given to you is pure water?
Answer: This can be confirmed by the following experiments :
- Filter the colourless liquid through a very fine filter paper. If no residue is left on the filter paper, this means that the liquid is pure water and has no suspended inpurities present in it.
- Evaporate the colourless liquid in a china dish or beaker. In case no residue is left, this means that it is pure water and has no dissolved impurities present in it.
- Determine the boiling point of pure liquid. If it comes out to be nearly 373 K (100°C), this means that the pure liquid is water.
Q7. Which of the following materials fall in the category of a “pure substance”?
(a) Ice
(b) Milk
(c) Iron
(d) Hydrochloric Acid
(e) Calcium oxide
(f) Mercury
(g) Brick
(h) Wood
(i) Air
Answer: The following materials fall in the category of a “pure substance”:
(a) Ice
(c) Iron
(d) Hydrochloric acid
(e) Calcium oxide
(f) Mercury
Q8. Identify the solutions among the following mixtures:
(a) Soil
(b) Sea water
(c) Air
(d) Coal
(e) Soda water
Answer: The following mixtures are solutions:
(b) Sea water
(c) Air
(e) Soda water
Q9. Which of the following will show the “Tyndall effect”?
(a) Salt solution
(b) Milk
(c) Copper sulphate solution
(d) Starch solution
Answer: Tyndall effect is shown by colloidal solution. Here milk and starch solution are colloids therefore milk and starch solution will show Tyndall effect.
Q10. Classify the following into elements, compounds and mixtures:
(a) Sodium
(b) Soil
(c) Sugar solution
(d) Silver
(e) Calcium carbonate
(f) Tin
(g) Silicon
(h) Coal
(i) Air
(j) Soap
(k) Methane
(l) Carbon dioxide
(m) Blood
Answer: Elements: Sodium, Silver, Tin and Silicon.
Compounds: Calcium carbonate, Methane and carbon dioxide.
Mixtures: Soil, Sugar, Coal, Air, Soap and Blood.
Q11. Which of the following are chemical changes ?
(a) Growth of a plant
(b) Rusting of iron
(c) Mixing of iron fillings and sand
(d) Cooking of food
(e) Digestion of food
(f) Freezing of water
(g) Burning of candle
Answer: The following changes are chemical changes:
(a) Growth of a plant
(b) Rusting of iron
(d) Cooking of food
(e) Digestion of food
(g) Burning of candle
We Think the given NCERT Solutions for class 9 Science book Chapter 2 Is Matter Around Us Pure with Answers Pdf free download will assist you. If you’ve got any queries regarding CBSE Class 9 Science Is Matter Around Us Pure NCERT Solutions with Answers, drop a comment below and that we will come back to you soons.
